In
my previous commentary on the sex determination of Tokudaia spiny rats dated May 21, 2012, I mentioned that Amami and
Tokunoshima spiny rats (T. osimensis
and T. tokunoshimensis) have no Y
chromosome and no Sry gene and their
sex chromosome type is XO in both males and females and that the gene contents
of the male X and the female X chromosomes must be different. However,
examining ten genes that are related to sex determination, Kuroiwa et al. (1)
concluded that the two chromosomes contain the same set of genes. This suggests
that my statement was incorrect (A. Kuroiwa, personal communication).
However,
this does not necessarily exclude the possibility that there may be some
sex-determining genes located on the X chromosomes. This possibility is
supported to some extent by the mechanism of formation of XO zygotes. Because
both males and females have the XO chromosome type, they should produce the
gametes with one X or no X chromosome in both sexes. Therefore, the offspring may
have genotypes XX, XO, and OO with the probabilities of ¼, ½, and ¼,
respectively. However, genotypes XX and OO are not observed, so that they must
be lethal. Why is the genotype XX lethal? If the male X (Xm) and the
female X (Xf) are identical and normal, the genotype XmXf
should be viable and be able to establish the XX genotype in the population. One
possible explanation for the absence of XX (or XmXf) and
the persistence of only XO could be that Xm and Xf carry
the male and the female-determining genes which are incompatible with each
other and therefore XmXf individuals will die. Of course,
this is an overstretched interpretation at present, and we need to study the
genes located on the male X and the female X chromosomes more extensively.
Kuroiwa
et al. identified copies of Cbx2
genes on two autosomal chromosomes in each of the two Tokudaia species, and the number of copies was higher in males than
in females. One of the two chromosomes is linked to the Sox9 gene, as in the case of other mammalian species, and therefore
the Cbx2 gene located on this
chromosome appears to be the original one, and other Cbx2 genes located on the other chromosome are considered to be the
genes transferred from the original chromosome and then duplicated. Kuroiwa et
al. speculated that these duplicate Cbx2
genes are responsible for testis formation because the Cbx2 genes are known to suppress ovary development in humans and
mice. Kuroiwa (personal communication) then suggests that the new chromosome
carrying more Cbx2 genes has become a
neo-Y chromosome and the homologous chromosome carrying a smaller number of Cbx2 genes has become a neo-X
chromosome.
This
is an interesting suggestion, but it is not without deficiencies. First, there
is no proof that a larger number of Cbx2
copies is really responsible for sex determination. Second, there must be a
recombination reduction in the duplicate gene region to keep the duplicate
genes together. Otherwise, the neo-Y chromosome cannot be isolated from the neo-X
chromosome (2). Once no recombination system evolves in this region, lethal
mutations are expected to accumulate particularly in small populations (3). At
the present time, no such evidence seems to exist.
Of
course, identification of the sex determination gene in these species would be very
difficult because the population size of Amami and Tokunoshima spiny rats is
very small and protected by the Japanese government. Therefore, one cannot get even
small samples easily. Nevertheless, it would be important to clarify the
evolutionary mechanism of the XO/XO type, because this would give some insight
into the general pattern of Y chromosome evolution.
 |
|
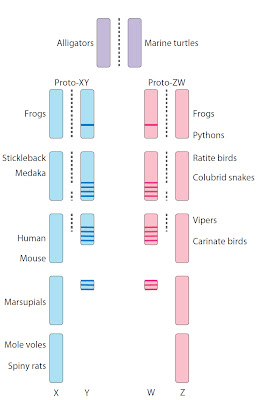
Fig.1.
Degeneration of the sex-specific element (Y or W) from an original autosome (purple) to form more or less
differentiated XY (blue) or ZW (pink) sex chromosome pairs. Degraded Y
chromosomes harbor male-specific genes (blue
lines) and W harbor female-specific genes (pink lines). Examples of vertebrate species that exhibit this
level of differentiation of XY or ZW chromosomes are given on the left or
right, respectively. Dotted lines represent pairing and recombination. From Graves (5).
|
Jennifer
Graves (4; 5; 6) has repeatedly argued that the human Y chromosome is subject
to a high rate of deleterious mutation and appears to become extinct in about
ten million years. It is known that only 50 out of the 1,500 genes that existed
in the original Y chromosome are surviving currently in the human chromosome.
She states: “Accelerated degeneration of the Y chromosome is found in 5 – 15%
of severely infertile men whose infertility is caused by wholesale deletion of
parts of this chromosome.” She envisages that once the Y chromosome is lost, a
new set of sex chromosomes like those of Tokudaia
spiny rats must be generated.
In
principle, I disagree with her, because strong purifying is operating for
keeping all Y chromosome genes required for male fertility and formation of
male phenotype. However, if contraception practice becomes more popular and
each married couple produces a smaller number of offspring, the purifying
selection will be weaker. It is then possible that infertile men increase and
the probability of decay of the Y chromosome genes may be enhanced. In this
case the formation of a new sex determination system similar to that of Tokudaia could be one solution. However,
because humans can control their evolution, it is unlikely for this event to
happen.
References
1.
Kuroiwa A, Handa S, Nishiyama C, Chiba E, Yamada F, Abe S, and Matsuda Y. 2011.
Additional copies of CBX2 in the genomes of males of mammals lacking SRY,the Amami spiny rat (Tokudaia osimensis) and the Tokunoshima spiny rat (Tokudaia tokunoshimensis). Chromosome Res 19:635-644.
2.
Nei, M. 1969. Linkage modification and sex difference in recombination. Genetics
63:681-699.
3.
Nei, M. 1970. Accumulation of nonfunctional genes on sheltered chromosomes. Am.
Nat. 104:311-322.
4.
Aitken RJ, and Marshall Graves JA. 2002. The future of sex. Nature 415:963.
5.
Graves JAM. 2008. Weird animal genomes and the evolution of vertebrate sex and sex chromosomes. Ann Rev Genet 42:565-586.
6.
Griffin DK. 2012. Is the Y chromosome disappearing?--both sides of the argument. Chromosome Res 20:35-45.
No comments:
Post a Comment